
Re-write the partial structural formula showing how these atoms are bonded to the carbon backbone (skeleton). Step 2: Identify any functional groups indicated by parentheses or any halogen atoms in the condensed structural formula, or semi-structural formula. Re-write the formula showing these branches (side-chains) as 2-dimensional structures Step 1: Identify any branches or side-chains in the condensed structural formula, or semi-structural formula. The new condensed structural formula, or semi-structural formula, is therefore (CH 3) 2CHClĬonverting a Condensed Structural Formula to a 2-Dimensional (full display) Structural Formula We then enclose the CH 3 groups in paretheses, (CH 3) and use the subscript number 2 to indicate that there are two of these groups attached to the same carbon atom, (CH 3) 2 It is possible to condense this structural formula even further by recognising that there is a central carbon atom to which both CH 3 groups are attached. Write the symbol for this chlorine atom to the right of the carbon atom of the backbone to which it is attached :Ĭondensed Structural Formula, or semi-structural formula, is CH 3CH ClCH 3 Step 6: This molecule contains one halogen atom (chlorine, Cl): Write the condensed structural formula, or semi-structural formula, for propane ( molecular formula C 3H 8) which has the 2-dimensional structural formula shown below:
Line structure chemistry calculator free#
No ads = no money for us = no free stuff for you! Condensed Structural Formula Worked Example: Short Straight Chain Hydrocarbons (d) write this group to the right of the carbon atom of the backbone at the position it is located. (c) use a subscript number to indicate the number of times this group occurs at that position (b) use parantheses (round brackets) to enclose groups of atoms attached to the chain (a) condense the structure as for the carbon backbone Step 6: If the molecule contains branches or side-chains: Step 5: Remove single carbon-carbon covalent bonds from the backbone or skeleton (this step is not absolutely necessary). Step 4: Re-draw the formula by replacing the identified groups of atoms in the 2-dimensional structural formula with their condensed structure, or semi-structural formula. Step 3: Identify groups of atoms along the carbon backbone in the 2-dimensional structural formula Step 2: Identify the carbon backbone (or skeleton) and any branches or side-chains in the structural formula as well as any functional groups such as halogen (group 17) atoms. Step 1: Draw the 2-dimensional structural formula for the molecule To write the condensed structural formula, or semi-structural formula:.For the sake of clarity, double and/or triple covalent bonds between carbon atoms are usually shown in the condensed structural formula, or semi-structural formula.If groups of atoms are attached to a chain, then parentheses (round brackets) are used to enclose groups attached to a chain.If there is repetition of a group in a chain, then brackets are used to indicate the repetition of a group in a chain (1)Ī subscript number written to the right of the final bracket tells us how many times this group of atoms is found in this position in the molecule.When the formula is written in a line with covalent bonds shown, it is also referred to as a linear formula. In a condensed structural formula, or semi-structural formula, covalent bonds are not always shown and atoms of the same type bonded to one another are grouped together.A condensed structural formula may also be referred to as a semi-structural formula.A condensed structural formula is a more compact way of drawing the structural formula of a molecule.A structural formula shows us how the covalent bonds are arranged around the atoms in a molecule.A Lewis Structure (electron dot diagram) shows us how the bonding and non-bonding valence electrons are arranged around the atoms.A molecular formula tells us how many atoms of each element make up the molecule.
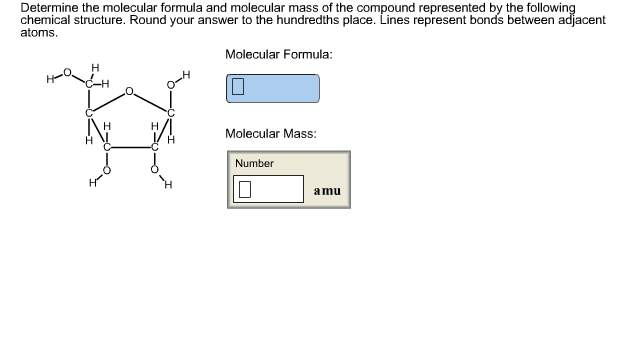
You need to become an AUS-e-TUTE Member! Condensed Structural Formula Chemistry Tutorial Key Concepts Want chemistry games, drills, tests and more?

Condensed Structural Formula Chemistry Tutorial More Free Tutorials Become a Member Members Log‐in Contact Us
